News
BHF Fellowship Awarded to Explore the Hidden Role of Placental Blood Vessels in Pregnancy and Fetal Heart Health
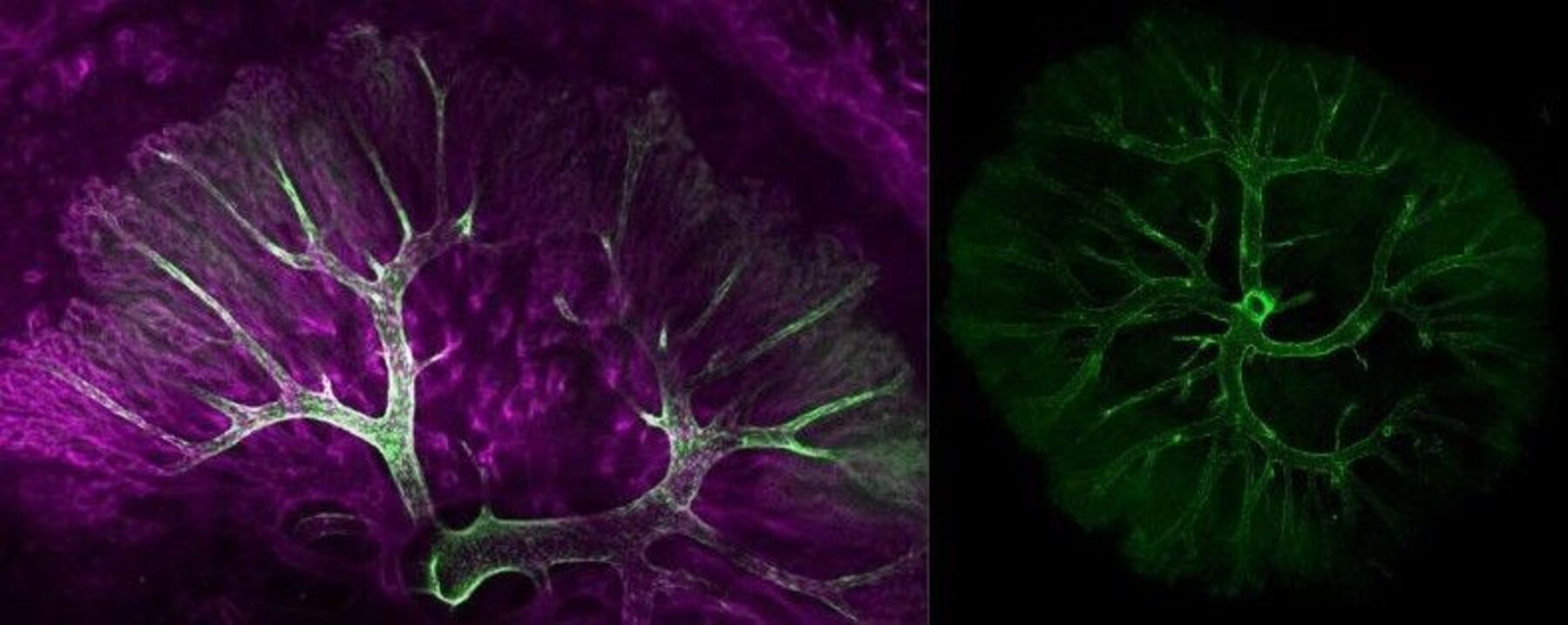
We’re thrilled to congratulate Dr Jacinta Kalisch-Smith on receiving a prestigious 5-year Intermediate Fellowship from the British Heart Foundation (BHF). Her research will shine a light on a part of pregnancy health that often goes overlooked: the blood vessels in the placenta, and how they may contribute to congenital heart defects and miscarriage.
Placental blood vessels play a vital role in supporting fetal growth and development, yet remarkably little is known about how they form or how their dysfunction might affect outcomes during pregnancy. Dr Kalisch-Smith’s fellowship aims to change that by uncovering the early origins of placental vasculature and how disruptions in this network could impact heart development in the fetus.
Investigating the placenta–heart connection
“My fellowship will understand the molecular mechanisms of placental blood vessel formation from their earliest progenitor cell populations in mice and humans,” says Dr Kalisch-Smith, “and how they affect or respond to pathologies of pregnancy, including miscarriage and congenital heart defects.”
While studies in mice have revealed some of the mechanisms behind placental vascular development, these insights are still in their infancy, especially when it comes to humans. The specific vascular network Jacinta is investigating, which connects the placenta to the fetus via the umbilical cord, has received very little attention in past research.
Yet, we already know this system is sensitive to both maternal environmental changes and fetal genetic factors. And evidence from mouse models suggests a strong two-way relationship: placental defects can lead to heart defects, and vice versa. However, this has never been confirmed in humans before birth, a key gap Dr Kalisch-Smith hopes to address.
"In addition, although we know mouse placental vascular development goes through a series of essential steps throughout gestation that must be completed for normal development, we don't know if they also occur in humans. This could have big implications on how we think about miscarriage, and its causes, via how the embryo-placental circulation is affected in early pregnancy."
A dual approach: mouse models and human tissue
Dr Kalisch-Smith 's research will combine mouse genetic models and human tissue from early pregnancy to understand how these blood vessels form in normal and pathological pregnancies, with a focus on miscarriage and congenital heart disease.
“I will be investigating placental-heart interactions in both mouse models and humans. In mice, we have the benefit of making new genetic tools to delete genes just from the placental vessels. I will be deleting genes from both arteries and veins with these new tools to see what types of heart defects are affected."
In humans, Dr Kalisch-Smith's research will investigate what and when placental vascular defects emerge during gestation with a focus on the first and second trimesters.
Targeting key gene pathways
Dr Kalisch-Smith's work also focuses on a fascinating group of genes known as imprinted genes, which can 'sense' the maternal environment and make changes to placental development to ensure healthy fetal growth. Some of these genes are strong candidates for influencing vascular formation, even though their exact roles are not yet known.
“I have a lot of good candidate genes to investigate, many without known roles in vascular formation, which is really exciting,” she says.
Dr Kalisch-Smith's interest in this field began during her PhD at the University of Queensland, Brisbane, Australia, where she studied how alcohol exposure very early in pregnancy, around two weeks from the time of conception, could affect placental formation.
“Here, I found alcohol caused knock-on effects to placental formation throughout gestation, including to the placental vasculature, which was really remarkable considering there wasn't a placenta formed at the stage of exposure. Considering how little we know about placental blood vessel formation, I am now in the best place to research this at the IDRM, surrounded by many labs here with a strong cardiovascular development focus, and the wider cardiovascular community in Oxford."
Towards prevention and early detection
Understanding how placental blood vessels develop and identifying when they show signs of dysfunction could pave the way for new biomarkers, molecules that could be detected in a mother’s blood to monitor placental health.
“By understanding what genetic pathways they (placental blood vessels) use, we can develop therapies to boost their formation and lessen the impacts of congenital heart defects and other common pregnancy pathologies like fetal growth restriction or stillbirth, which also implicate placental vascular defects,” Dr Kalisch-Smith says.
Her fellowship could also uncover which maternal environmental factors, such as nutritional deficiencies, disrupt placental vascular development. If identified, these factors could potentially be addressed through targeted interventions like pre-pregnancy supplements, much like how folate is used to prevent neural tube defects.