News
A new study challenges a potential gene-activating mechanism using antisense oligonucleotides
A new study from the Roberts Group, Ahlskog et al., published in Molecular Therapy: Nucleic Acids, challenges the therapeutic potential of a mechanism previously thought to upregulate protein expression in specific genes.
The paper, "uORF-targeting steric block antisense oligonucleotides do not reproducibly increase RNASEH1 expression," focuses on steric block antisense oligonucleotides (ASOs) targeting a regulatory element in gene transcripts that inhibits protein production.
The development of ASOs has revolutionised molecular medicine by offering targeted approaches to modulate gene expression. Steric block ASOs, in particular, have been successfully directed at different gene targets to create therapies for various diseases. Multiple steric block ASOs have already been approved for the treatment of Duchenne muscular dystrophy and spinal muscular atrophy.
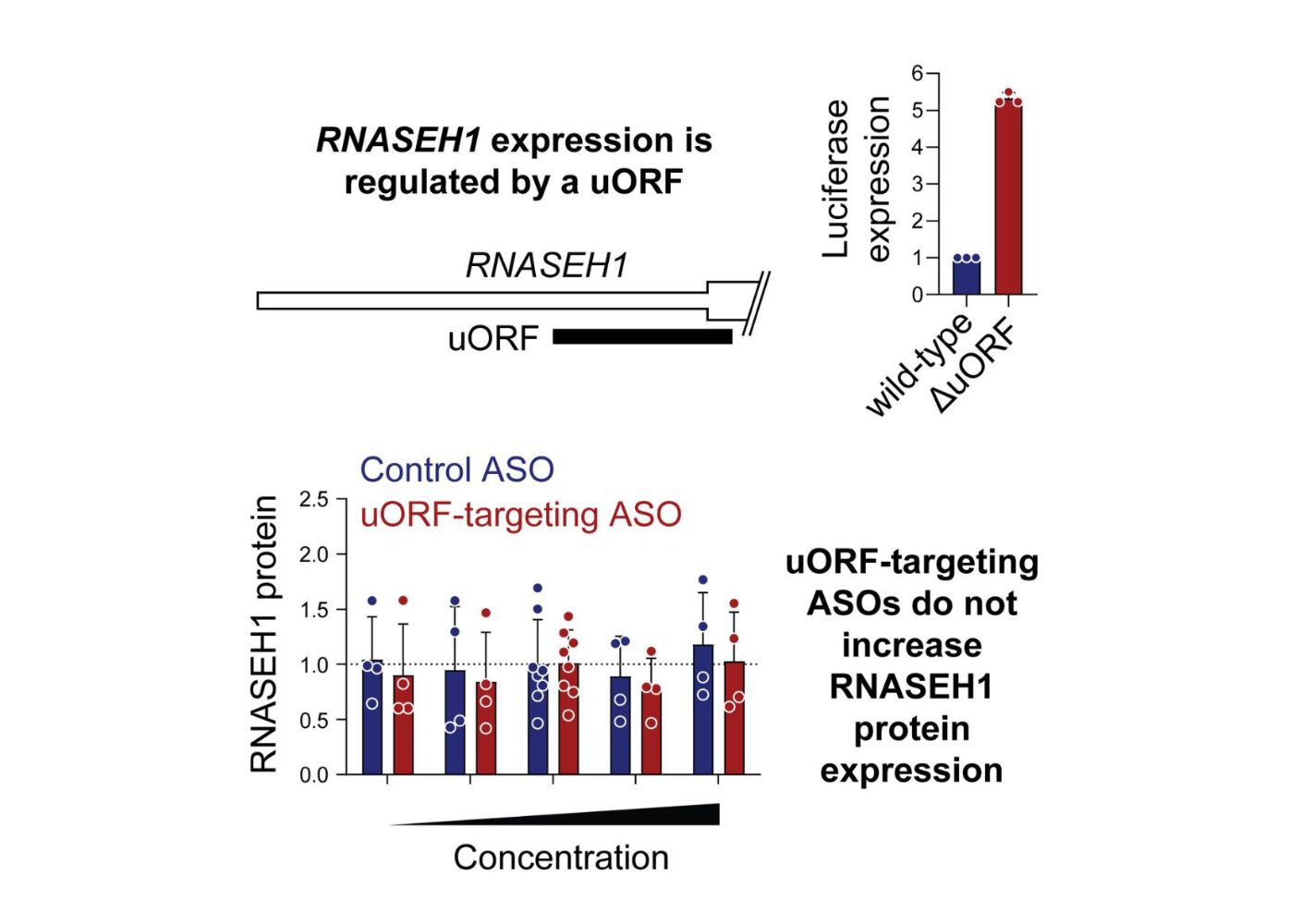
Targeted activation of specific genes has been reported as one potential use for steric block ASOs, as previously described in a 2016 study by Liang et al.
The Liang et al. study reported successful protein upregulation by using ASOs to target upstream open reading frames (uORFs) — regulatory elements that typically suppress translation of the primary open reading frame (pORF). If such a therapeutic modality proved reliable, it could be applied to diseases where increasing specific protein levels could offer therapeutic benefits, such as certain genetic disorders.
Researchers from the University of Oxford attempted to reproduce the findings reported by Liang et al. for RNASEH1 protein upregulation. The team used three uORFs-targeting ASOs reported by Liang et al. and conducted a series of experiments to evaluate whether these ASOs could upregulate RNASEH1 protein expression across a range of concentrations (25–300 nM).
Contrary to the original findings, the authors observed no significant increase in RNASEH1 protein levels. In some cases, they noted a decrease in protein expression, which suggests potential off-target effects or unanticipated consequences of ASO binding. These results remained consistent across multiple experimental setups.
The study concluded that previously described steric block ASOs that target uORFs do not reproducibly activate RNASEH1 (ribonuclease H1) expression. This challenges earlier findings that proposed uORF-targeting ASOs as a promising mechanism to upregulate gene expression.
While the modulation of uORFs remains an innovative and theoretically promising strategy for the development of genetic therapies, the study by Ahlskog et al. highlights barriers to its practical application. Further research into ASOs is needed to determine whether alternative ASO design or delivery strategies can achieve consistent gene activation.
Associate Professor Thomas C. Roberts said: " We are highly interested in uORFs, and how we can utilise these for therapy. We were hoping that direct targeting of uORFs, as was reported previously, could be used to upregulate therapeutically relevant genes. However, in our study we show that the situation is more complex, which has motivated us to think more creatively about uORF targeting – an area of ongoing research in my group."
This study is part of a much wider effort to target uORFs for upregulating therapeutic genes and was funded by a grant from Great Ormond Street Hospital Sparks Fund.